AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic radius of flourine12/27/2023 97–100 The main advantages of these methods are easy implementation and computational efficiency. This can be done by reducing the van der Waals’ radii of atoms, or using a softer energy function to allow some degree of atomic clash, or allowing certain atoms to completely penetrate each other. Wade, in Comprehensive Medicinal Chemistry II, 2007 4.09.4.4.1 Implicit modelsĪ simple way to represent a limited degree of flexibility is to use a soft representation of molecules. 
Similarly, in case of CD578 the alkyne functionality is relatively resistant to metabolic oxidation due to the presence of the adjacent trifluoromethyl groups ( Figure 9).ĭ. 44 In the case of the WY1113, the two CF 3 moieties on the C 25 are able to inhibit the hydroxylation at C 24. 43 The nonsteroidal analogs of vitamin D 3―WY1113 and CD578―with two CF 3 groups on the side chain not only are metabolically stable to oxidative degradation, but also are superior agonists for the vitamin D receptor, as compared to their nonfluorinated analogs and 1,25-dihydroxyvitamin D 3. 42 However, strategic replacement of the C 24-hydrogens by two fluorine atoms results in greater metabolic stability. The 1,25-dihydroxyvitamin D 3 is prone to hydroxylation at the C 24 carbon by cytochrome P24 (24-hydroxylase) family of enzymes, which leads to its rapid oxidative degradation ( Figure 9). Further, due to the large electron-withdrawing inductive effect, the neighboring C–H bonds also are relatively polar and their oxidative stability is enhanced. The relatively greater C–F bond strength imparts greater metabolic stability to the organofluorine pharmaceuticals when the relatively labile C–H bond is replaced by the C–F bond. 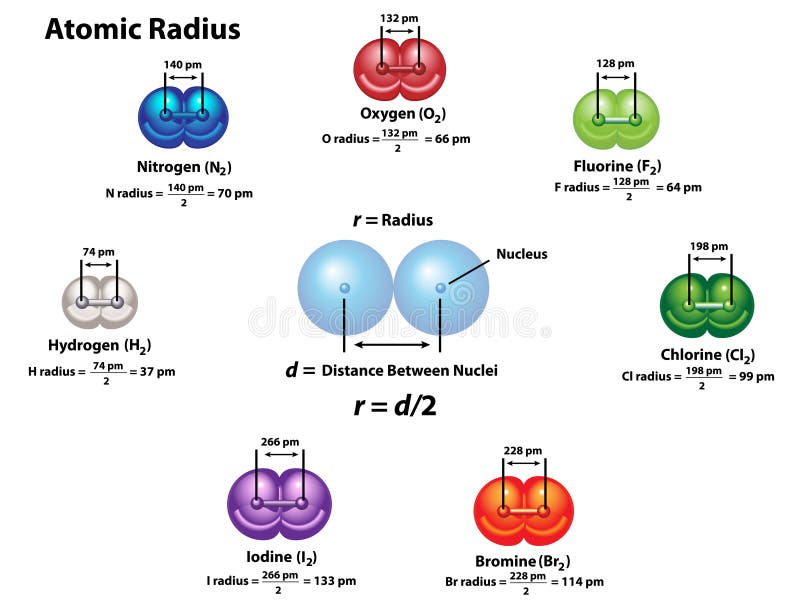
It is interesting to note that, in general, as shown by the above examples, a strategic substitution of aryl hydrogen by fluorine in the lead compounds can lead to superior drug candidates with improved bioavailability and potency. 41 Substitution of the aryl fluorine by a methyl group helped to decrease the metabolic stability to an acceptable level of 3.5 h for celecoxib. For example, the plasma half-life of a drug candidate with a fluorophenyl moiety, SC-57666, an analog of celecoxib (a COX-2 inhibitor used for treatment of rheumatoid arthritis and osteoarthritis), is 221 h in rats. 40 On the other hand, sometimes fluorine exerts undesirably high metabolic stability in drug candidates. 
Similarly, the strategic incorporation of fluorine into the aromatic ring results in increased potency and efficacy in vivo for linezolid, which is an FDA-approved antibacterial drug to treat infections caused by vancomycin-resistant bacteria ( Figure 8). Structures of ciprofloxacin, linezolid, SC-57666, and celecoxib.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
